Elements in period 4 have the forth energy levelĪs the outer most level to be occupied by electrons. Similarly atoms in groups 2-8 have 2,3,4,5,6,7 or 8 electrons respectively.Īn element in period 2 has the second energy level as the last level toīe occupied by electrons. These elements do not readily react with other elements.Įlements in group 4, 5, 6,7 and 8 are nonmetals.Įlements in group 1 have ONE electron in their outer most energy level. Is there a pattern?Įlements in group 8 exist as gases and are very stable. Now compare the reactivity of calcium and magnesium with water. Compare the reactivity of sodium and potassium with water. Notice how reactivity of metals increases as we go down a group. Potassium reacts faster than sodium to produce hydrogen gas.Įlements in group 2 also react with water to form hydrogen gas but are not as reactive as group 1 elements. Sodium reacts slowly with water to produce hydrogen gas. 
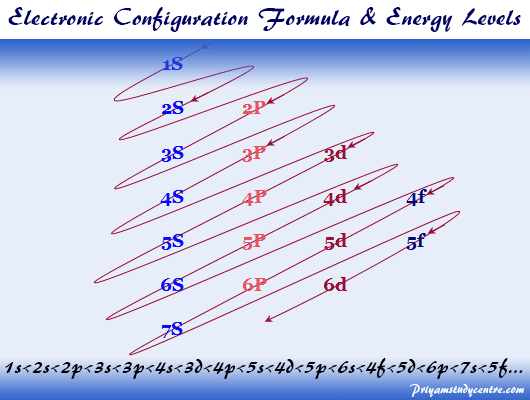
These metals react readily with water to produce hydrogen gas. Elements in group 1 are very reactive metals, with the exception of hydrogen Can you explain the trend in atomic radius?Īll elements in a particular group share similar properties. Look at the electronic configuration of each atom. Elements in the same group display similar chemical properties and trends emerge as we go along a period and down a group.Ĭlick to see how size of atoms changes across a period and down a group. The table is divided into groups(columns) and periods(rows). The modern periodic table has elements arranged in order of electronicĬonfiguration. Recognise that elements can be grouped together according to atomic mass. Mendelev was a Russian scientist who was first to So those are the exceptions the main exceptions that you'll see when dealing with lifetime configuration.The periodic table is a way of organizing known elements into groups with So, again, I'm going to make this argon just make it for myself 4s1, 3d10 and again you might see it as argon 3d10, 4s1 same exact thing. That way this d orbital or d sublevel be completely filled which is very stable versus the s orbital will be halfway filled. Okay so anytime that it ends in d9 we're going to fix it up a little bit, same exact way we're going to take an electron from the 4s and we're going to move it over to the 3d. So let's make the electron configuration for that 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d9 from here on out anytime you see d9 we're going to change it. So you might see either one but they're the same.ĭown here the other exception that you're going to see is copper, or anything along copper which we'll talk about. So this is talking about the order of energy you might see it also looking like this, this is just doing it in order of number 3 comes before 4 but they're exactly the same thing, they're depicting the exact same thing, nothing different about them. 
You might see this is the diagram or the electron configuration that you're going to see and this is actually higher in energy than this. So instead we're going to write actually we're going to make this argon as we noted before in the noble gas configuration and we're going to make this 4s1, 3d5 this is halfway filled which is pretty stable and this is much more stable being half way filled rather than being the d4. So what we're going to do, we're going to, that should be a 2 there, sorry, and we're going to take one of the electrons in the 4s orbital and move it over to the 3d orbital. Right now we're 1 electron short of it being halfway filled. The d sublevel is more stable when its either half full all the orbitals are filled with at least 1 electron or completely filled. At the end of each row, a drastic shift occurs. If we're going to make this short hand and make the electron configuration for this we would make this 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d4 okay from now on every time you see 3d4 you're going to change it, we do not like 3d4. As one moves from left to right in a row of the periodic table, the properties of the elements gradually change. Chromium is a transition metal and it has 24 electrons and here is the orbital diagram. Okay alright so let's talk about the exceptions you're going to see when you're dealing with electron configuration there's going to be a few around but we're going to talk about the main ones you're probably going to see in class. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
